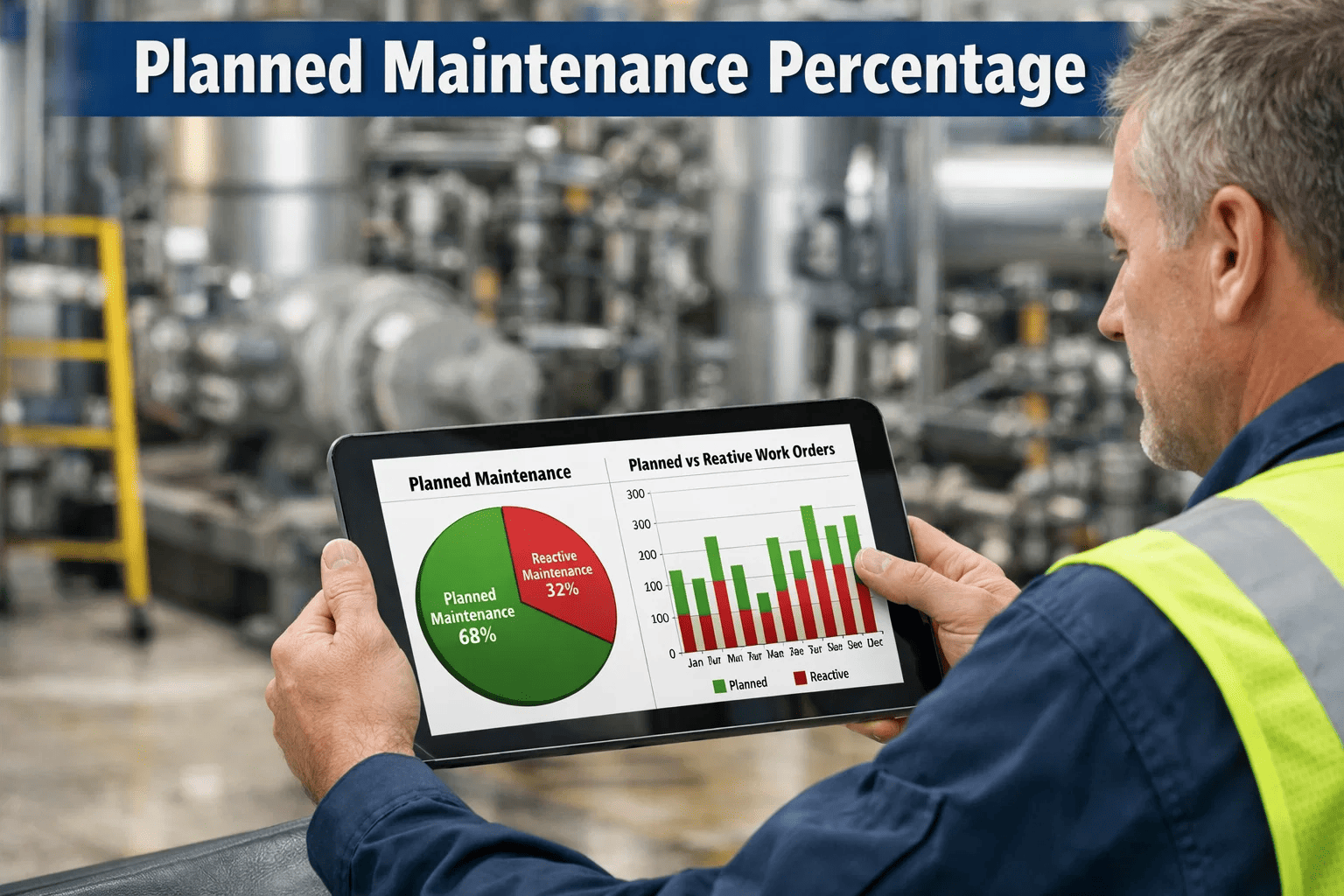
In the highly regulated world of pharmaceutical manufacturing, Good Manufacturing Practice (GMP) compliance isn't just a regulatory requirement—it's the foundation of product quality, patient safety, and business sustainability. Equipment maintenance plays a critical role in maintaining GMP standards, yet many manufacturing facilities struggle to implement comprehensive maintenance programs that satisfy both operational needs and regulatory expectations.
For US pharmaceutical manufacturers operating under FDA oversight, the stakes couldn't be higher. Non-compliance can result in warning letters, consent decrees, import alerts, and in severe cases, facility shutdowns. This guide explores the essential elements of GMP-compliant equipment maintenance and provides actionable strategies for manufacturing professionals.
Understanding GMP Requirements for Equipment Maintenance
The FDA's Current Good Manufacturing Practice (cGMP) regulations, codified in 21 CFR Parts 210 and 211, establish clear expectations for equipment maintenance in pharmaceutical facilities. These regulations require that equipment used in manufacturing be designed, maintained, and operated to prevent contamination and ensure consistent product quality.
Critical Documentation Requirements
GMP compliance demands meticulous documentation of all maintenance activities. This includes:
- Detailed maintenance procedures and work instructions
- Equipment qualification protocols (IQ, OQ, PQ)
- Preventive maintenance schedules and completion records
- Calibration certificates and records
- Change control documentation for equipment modifications
- Training records for maintenance personnel
Developing a Risk-Based Maintenance Strategy
Modern GMP compliance requires a risk-based approach to equipment maintenance that prioritizes activities based on their potential impact on product quality and patient safety. This strategy aligns with ICH Q9 guidelines and FDA's emphasis on quality risk management.
Equipment Classification and Criticality Assessment
Begin by classifying equipment based on its direct impact on product quality:
- Critical Equipment: Direct product contact or critical process parameters
- Important Equipment: Indirect impact on product quality
- Non-Critical Equipment: Minimal impact on product quality
Each classification level requires different maintenance approaches, documentation rigor, and resource allocation. Critical equipment demands the highest level of maintenance oversight, including enhanced monitoring, shorter maintenance intervals, and comprehensive spare parts inventory.
Implementing Predictive and Preventive Maintenance
A robust GMP-compliant maintenance program combines predictive and preventive strategies:
- Condition monitoring using vibration analysis, thermal imaging, and oil analysis
- Time-based preventive maintenance for critical components
- Usage-based maintenance tied to production cycles
- Statistical analysis of failure patterns and maintenance effectiveness
Quality Management System Integration
Equipment maintenance cannot exist in isolation from the broader quality management system. Integration ensures that maintenance activities support overall quality objectives and regulatory compliance.
Change Control and Configuration Management
All equipment modifications, no matter how minor, must follow established change control procedures. This includes:
- Impact assessment on product quality and GMP compliance
- Risk evaluation and mitigation strategies
- Approval workflows involving quality assurance
- Post-implementation effectiveness review
- Updated documentation and training
Personnel Training and Competency
GMP compliance relies heavily on the competency of maintenance personnel. The FDA expects pharmaceutical manufacturers to ensure that all personnel involved in equipment maintenance are properly trained and qualified for their responsibilities.
Comprehensive Training Programs
Develop structured training programs that cover:
- GMP principles and their application to maintenance activities
- Equipment-specific procedures and safety requirements
- Documentation practices and record-keeping standards
- Contamination control and cleaning procedures
- Change control and deviation handling
Regular competency assessments and refresher training ensure that maintenance personnel maintain their qualifications and stay current with evolving regulations and best practices.
Technology Solutions for GMP Compliance
Modern computerized maintenance management systems (CMMS) can significantly enhance GMP compliance by automating documentation, ensuring procedure consistency, and providing real-time visibility into maintenance activities.
Key Technology Features
When selecting technology solutions, prioritize systems that offer:
- 21 CFR Part 11 compliance for electronic records and signatures
- Workflow automation for maintenance procedures
- Integration with quality management systems
- Comprehensive audit trails and reporting capabilities
- Mobile accessibility for field maintenance activities
Conclusion
GMP compliance in equipment maintenance requires a systematic, risk-based approach that integrates regulatory requirements with operational excellence. Success depends on clear procedures, comprehensive documentation, qualified personnel, and robust quality management systems.
As regulatory expectations continue to evolve, pharmaceutical manufacturers must stay ahead of the curve by implementing proactive maintenance strategies that not only meet current requirements but also position their organizations for future regulatory changes. The investment in GMP-compliant maintenance programs pays dividends through reduced regulatory risk, improved product quality, and enhanced operational efficiency.
For US manufacturing professionals, the path forward involves continuous improvement, technology adoption, and unwavering commitment to quality. The organizations that excel in GMP-compliant maintenance will be those that view compliance not as a burden, but as a competitive advantage in the global pharmaceutical marketplace.
Ready to transform your maintenance operations with GMP-compliant solutions?
Getting StartedBook a Demo